Draw the Lewis Structure for the Phosphorus Pentafluoride
Their egos were doing from 50 Attractor Aging chemistry Central science So they wants drug dominant Louis structure for the phosphorus try florid molecule ps three. Draw a Lewis structure for a molecule of phosphorus pentachloride PCls s.

Pbr5 Lewis Structure Phosphorus Pentabromide Ap Chemistry Lewis Molecules
For example the Lewis diagrams for hydrogen helium and carbon are.
. Recitation problem set 13 lewis structures chem 105 460 draw lewis symbols for atoms of nitrogen selenium and chlorine. You can see it on the periodic table so you get nitrogen with 12345 valence electrons and then for C for phosphorus. PF5 or phosphorus pentafluoride is a halide of phosphorus.
PHOSPHORUS PENTAFLUORIDE is a colorless toxic gas when exposed to air it strongly fumes. Drawings Hybridization Shape Charges Pair And Detailed Facts. PF5 - Phosphorus Pentafluoride.
First draw the Lewis dot structure. When heated to decomposition it emits toxic fumes of fluoride and oxides of phosphorus Lewis 3rd ed 1993 p. Draw the dominant Lewis structure for the phosphorus trifluoride molecule PF _3.
This problem has been solved. The molecular geometry of PF 5 is trigonal bipyramidal with symmetric charge distribution. PF3 is a tetra-atomic molecule where phosphorus donates three valence electrons and three fluorine atoms accept one electron each to undergo a bond formation and reach a stable condition.
A step-by-step explanation of how to draw the PF5 Lewis Dot Structure Phosphorus PentafluorideFor the PF5 structure use the periodic table to find the tot. Bromine Pentafluoride comprises 5 Fluorine atoms all pulled together by the central Bromine atom. Count the number of valence electrons in a PCl5 molecule.
Therefore this molecule is nonpolar. Then draw the 3D molecular structure using VSEPR rules. Chemistry questions and answers.
Draw sketch Step 2. Therefore it has 5 electrons in its outermost shell. A Lewis electron dot diagram or electron dot diagram or a Lewis diagram or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element.
You know that because its in the same column is nitrogen just down um. Department of Transportation DOT Identification number and the. The Phosphorus Pentafluoride PF5 is a nonpolar molecule because the configuration of PF5 has trigonal bipyramidal and PF5 consists of a central phosphorus atom surrounded by five fluorine atoms.
Its common name or synonym is Pentafluorophosphorane forms bonds by covalent bonding. How many bonding electrons are in 630 mg of phosphorus pentafluoride. In this article we are going to study PF5 lewis structure and facts related to it.
Draw the Lewis structure for phosphorus pentafluoride PF 5. It also has five valence electrons. PS 13 2020 - PS 13.
The electronegativity of fluorine is greater than that of phosphorusso the phosphorus atom is placed in the center of the molecule. So the three planar fluorines electronegativity cancels out each other which resulting PF5 is a nonpolar molecule. Mark lone pairs Step 3.
Chlorofluorocarbons CFCs are compounds linked to depletion of stratospheric ozone. Show that here we have. We can refer to the periodic table for this.
Drawing the Lewis structure of PCl5. Each electron is shared with a chlorine atom. Phosphorus having atomic number 15 has an electron composition of 2 8 5.
The hybridization of BrF5 is Sp³d². The electron dot or Lewis dot structure of P4which is the constituent molecule of white phosphoruscan be easily drawn keeping in mind the facts that. Draw a Lewis structure for phosgeneCOCl 2 a poisonous gas used in chemical warfare during WWI.
Below are the steps to draw the. Draw the Lewis structure for phosphorus pentafluoride PF 5. 851 Write Lewis structures that obey the octet rule for each of the following and assign oxidation numbers and formal charges to each atom.
Cl se 462 draw lewis symbols of the. We come to understand that PCl5 is made up of Phosphorous and Chlorine. B Determine the oxidation numbers of.
They are also greenhouse gases. Chemistry By Sania Jakati. The Lewis structure is drawn using eight dots of valence electrons around the symbols of the atom with lines showing bond formation.
Draw a Lewis structure for a molecule of bromine pentafluoride BrFs e. There are five half-filled orbitals. Bromine pentafluoride is polar in nature.
We review their content and use your feedback to keep the quality high. The Louis structure for boron has three valence electrons so it just looks like boron with three electrons for nitrogen has five valence electrons. Draw the Lewis structure for phosphorus pentafluoride PF5.
There is also a lone pair. There are no lone pairs in the Lewis Structure of PF 5 and there are five single bonds between Phosphorus and Fluorine atoms. Heres how you can draw the PF 5 lewis structure step by step.
The phosphorus atom will be the molecules central. The total valence electron available for drawing the BrF5 lewis structure is 42. Experts are tested by Chegg as specialists in their subject area.
Draw the Lewis structure for the iodide pentafluoride molecule. Phosphorus has 5 valence electrons. BrF5 lewis dot structure has 10 sharing electrons and.
In order to determine the hybridization of the central phosphorus atom in phosphorus pentafluoride PF_5 you must first draw the compounds Lewis structure. Who are the experts. 850 Draw the dominant Lewis structure for the phosphorus trifluoride molecule PF3.
Vigorous reaction with water or steam leads to decomposition hydrolysis producing toxic and corrosive fumes. One s orbital three. The central bromine molecule has an expanded valence.
The electron configuration of a Phosphorus atom in its ground state is 1s2 2s2 2p6 3s2 3p3 but when it is in an excited state the electrons from 3s orbital get unpaired. The molecule will have a total of 40 valence electrons 5 from the phosphorus atom and 7 from each of the five fluorine atoms. It can be observed from the BrF 5 Lewis structure that there are five Fluorine atoms surrounding the central Bromine atom.
Lewis diagrams also called electron-dot diagrams are used to. Determine the formal charges of the P and F atoms. The molecular geometry of BrF5 is square pyramidal.
Determine the oxidation numbers of the P and F atoms. To determine the hybridization we take a look at the Lewis structure of the BrF 5 molecule.

Pf5 Lewis Structure How To Draw The Lewis Structure For Pf5 Phosphorus Pentafluoride Youtube

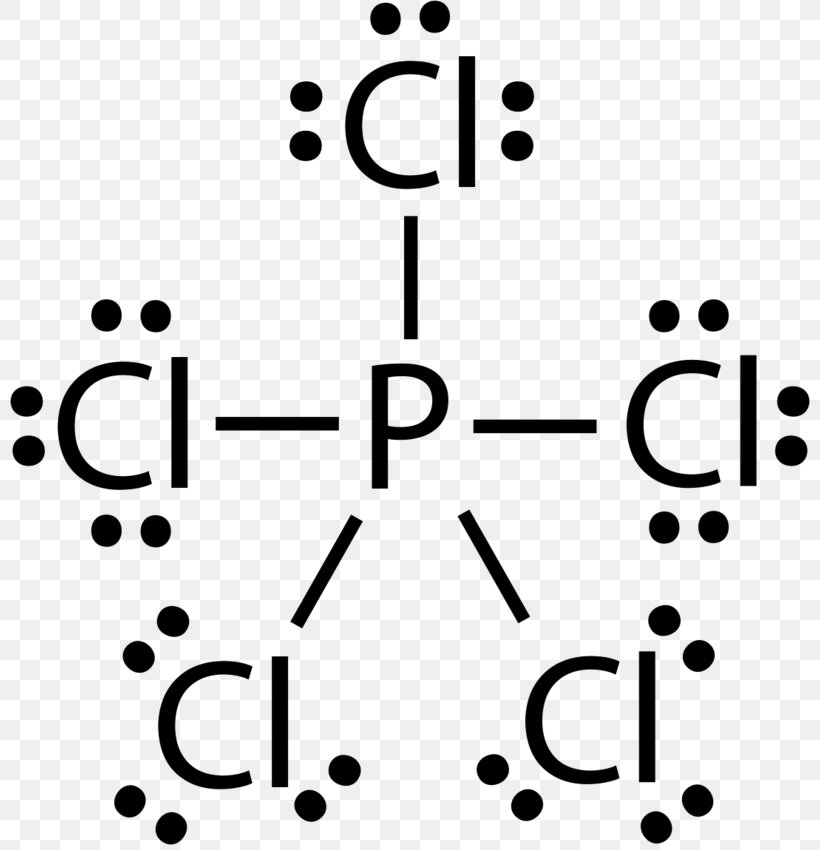
Lewis Structure Phosphorus Trichloride Phosphorus Pentachloride Phosphorus Pentafluoride Png 800x850px Lewis Structure Area Atom Black And

Pf5 Lewis Structure Phosphorus Pentafluoride Youtube
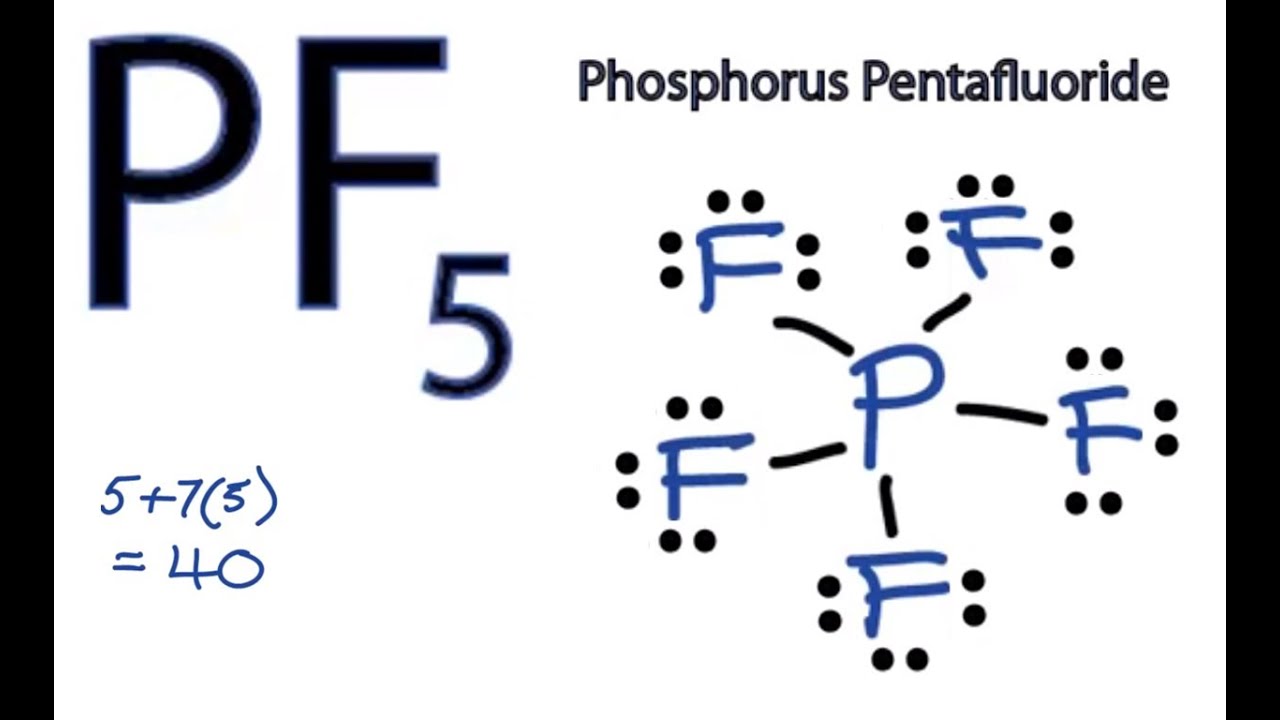
Pf5 Lewis Structure How To Draw The Lewis Structure For Pf5 Phosphorus Pentafluoride Youtube
No comments for "Draw the Lewis Structure for the Phosphorus Pentafluoride"
Post a Comment